Medicines Manufacturing Industry Partnership (MMIP)

The Medicines Manufacturing Industry Partnership (MMIP) represents the voice of medicines manufacturers in the UK. It was established jointly by the government and the biopharmaceutical industry in 2014 to ensure that the UK is recognised by the global medicines industry as a world-class advanced centre for medicines manufacturing.
The UK's medicines industry is one of the country's leading manufacturing sectors, and one of the UK’s highest value exports.
The Partnership aims to ensure that the UK continues to be an attractive place for this high-value industry to secure exports, provide high skilled job opportunities and contribute to solving the country's productivity puzzle.
The UK Life Sciences Vision recognised the sector’s high productivity and the MMIP is now an expert group under the direction of the Life Sciences Council.
MMIP Structure
Working closely with key government organisations including Innovate UK and the Office for Life Sciences (OLS), MMIP is continuously working to sustain the UK's leading position in medicines manufacturing technologies, including those related to advanced therapy medicinal products (ATMPs), leadership in sustainable medicines production, and continuous production and digitalisation.
MMIP’s vision:
In June 2023 the MMIP published ‘Follow the green, high-tech road: A path to UK growth, net zero and health resilience from innovation in medicines manufacturing’.
The report sets out a comprehensive 10-year plan to return the UK to its status as a world leader in advanced medicines manufacturing.
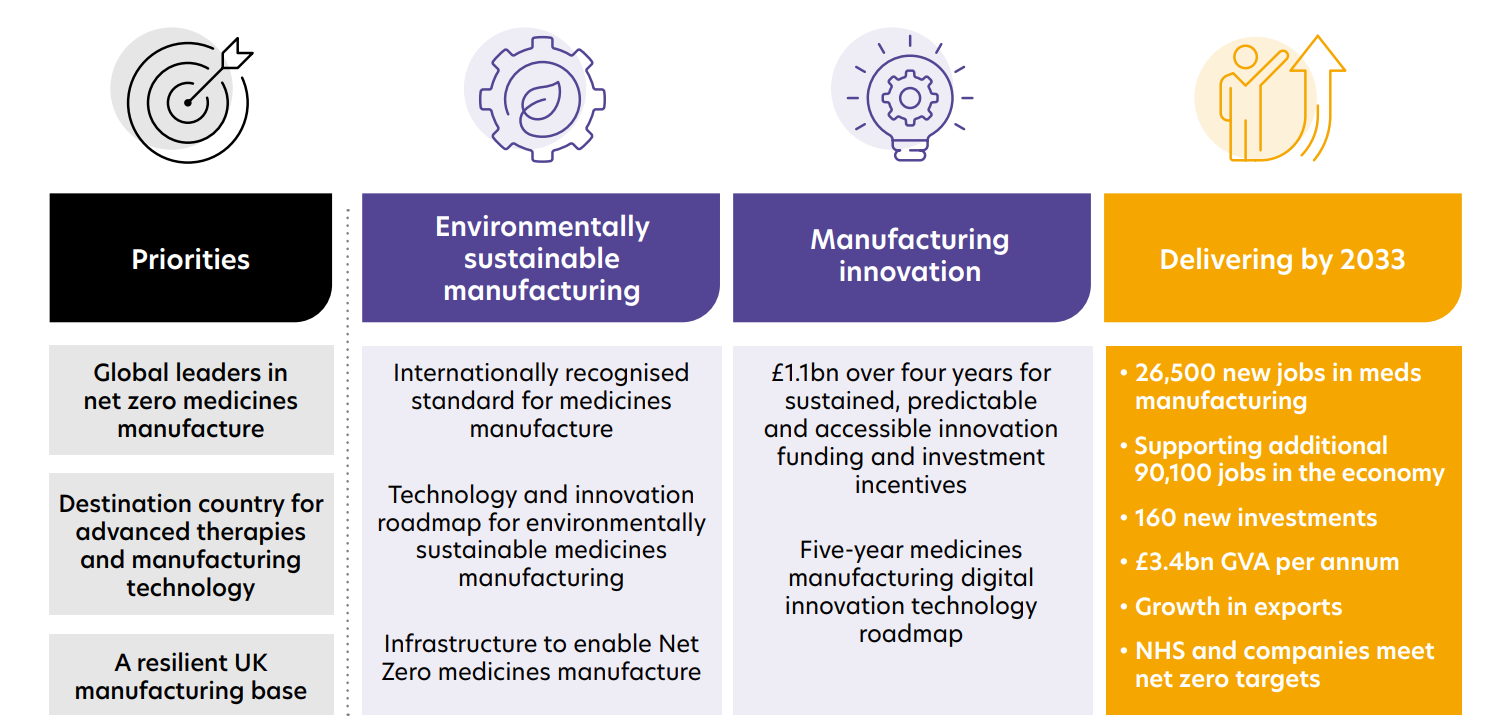
Download larger version of chart
Workstreams
To make progress on this shared agenda, the MMIP is structured into workstreams.
- the technology and innovation landscape
- the fiscal environment
- the regulatory environment
- the skills environment
- Advanced Therapies manufacturing
Support
The MMIP is supported by the ABPI, the BioIndustry Association (BIA), the Knowledge Transfer Network, and a range of leading biopharmaceutical manufacturers.
Contact Us
For more information please contact:
- Joe Edwards – Director of UK Competitiveness and Devolved Nations
- Bill Dawson - Head of Biologics and Devices Manufacturing at GSK - Chair of the Medicines Manufacturing Industry Partnership (MMIP).
Related news and blogs
-

Boost UK manufacturing to attract £15 billion new investment, says medicines manufacturing alliance
With the right policies in place, the UK can be the best global location for innovative and environmentally sustainable medicines manufacturing, according to a new report from the Medicines Manufacturing Industry Partnership (MMIP).
-

Medicines manufacturing has the potential to drive UK growth over the next 10 years
Recent successes in supplying high-value medicines and in advanced therapies manufacture must be built on, according to a cross-industry coalition.
-

Pfizer’s Brian Henry appointed head of UK medicines manufacturing partnership
The Medicines Manufacturing Industry Partnership (MMIP) represents the voice of medicines manufacturing in the UK.

The supply chain workstream seeks to deliver capable and connected infrastructure, building understanding of medicines manufacturing supply chains, identifying opportunities to maximise supply and fostering non-competitive sector collaboration.

The technology workstream seeks to understand and pursue the future needs of the medicines manufacturing industry.

The Knowledge Transfer Network (KTN) have created a tool which aims to provide a visual dynamic representation of the UK Medicines Manufacturing Innovation Landscape.

MMIP News and Publications

Events where the MMIP team will be presenting.

The regulatory environment workstream is trying to maximise innovative processes by highlighting existing regulatory flexibility and the positive UK regulatory landscape to industry.

As part of its programme of work, the Medicines Manufacturing Industry Partnership (MMIP) seeks to ensure that the UK has an adaptive workforce with relevant skills to sustain the future requirements of the UK medicines manufacturing industry.
