Executive summary
Rare diseases affect over 3.5 million people in the UK. While progress has been made in the diagnosis and treatment of rare diseases, there is still a significant amount of unmet need.
The UK Rare Diseases Framework sets out the government's ambition to improve care for people living with rare diseases by:1
1. Improving speed of diagnosis;
2. Increasing awareness of rare diseases amongst healthcare professionals;
3. Improved coordination of care; and
4. Improved access to specialist care, treatment, and drugs.
These priorities are a key part of addressing current challenges in rare diseases.
"The current landscape for treatment availability is really patchy and inequitable if you compare different conditions.[…] So we're quite keen to find ways to democratise the concept of highly specialised services."
Nick Meade, Genetic Alliance UK
This study explores the benefits that could be gained from improving treatment availability for rare diseases to individuals, the health system, and the economic growth of the nation. It looks at benefits associated with the discovery of new treatments for rare diseases, as well as a higher proportion of these innovative medicines being made available to people on the NHS.
The current landscape of rare diseases
▪ While individually rare, over 3.5 million people in the UK live with a rare disease, many of whom are children.2
▪ Currently only 5 per cent of rare diseases have an effective treatment.3
▪ Where treatments are licensed in Europe, only 35 per cent are fully accessible for NHS patients (inclusion on public reimbursement list).4
▪ Children with rare diseases are twice as likely to be admitted to hospital than those without a rare disease, and stay 5 times longer.5
▪ 70 per cent of rare disease patients and carers had to stop or reduce professional activity.6
Looking forward: How can the UK enhance access to rare disease treatments
Currently, only around 5 per cent of rare diseases have an approved medicine for prevention, diagnosis, or treatment. Continued innovation in the orphan medicines R&D environment could more than double this to 11 - 15 per cent.7
The UK can play a role in maximising the opportunities for this pipeline to come to fruition by continuing to maintain a supportive environment for research and development (R&D).
Innovations in basic and translational science have led to an increase in the understanding of rare diseases. For example, the rise in genomic testing has led to the identification of many more targets for medicine development. In England, the NHS 10 Year Health Plan sets out a clear ambition for increased application of genomics in treatment, research and prevention of disease, stating that by 2035, half of all healthcare interactions will be informed by genomic insights. If we were to realise this ambition, this could support research crucial to discovering the cause and eventually treatments for these conditions.
The clinical trials process is another key step in the approval of new medicines. Clinical trials for rare conditions face unique challenges as, by their very nature, they are only able to recruit a small number of participants who are geographically dispersed. Patients are often clinically heterogenous, meaning trials are more susceptible to variability of findings, impacting the study's scientific conclusions.
By virtue of involving fewer patients, these trials also produce less data and often don't have clear endpoints to measure, which creates added challenges for medicines regulators and the assessment of the cost effectiveness of a new treatment.
Basket trials, where one therapy is used for multiple diseases with shared characteristics are a novel approach to overcome some of these challenges. Personalised therapies often require small, well-controlled studies rather than traditional trial designs, and effectiveness and safety monitoring gathered as real-world evidence. For life-limiting conditions or serious conditions where there is unmet need accelerated approval processes are in place which accept a surrogate endpoint rather than clinical benefit.
In the UK clinical trials face many challenges including slow set-up times due to delays in site opening post regulatory approvals and decreasing recruitment in the NHS.8 Recruitment into UK industry trials has fallen for the fourth year in a row to only 19,092 in 2024/25. Only 3.4 per cent of all trial participants in England in 2024/25 were recruited to industry trials in the NHS.
These challenges are raising costs and lowering the efficiency of running commercial trials in the UK compared to European and international peers. The Government have committed to reducing trial set-up time to less than 150 days by end of March 2026 which could help reverse this trend and increase clinical trial activity so that more UK patients have the opportunity to access treatments.
In addition, the UK Life Sciences Sector Plan sets out an ambition that, by 2030, the UK will be one of the top three fastest places in Europe for patient access to medicines and MedTech. Looking across 2020–2023, the third highest full availability rate for orphan medicines in Europe was 50 per cent.
Looking forward: Benefits to the UK of improving access to rare disease treatments
In this study we explore the benefits of improved access to rare disease treatments to patients, the NHS, and the UK economy compared to today. The scope of this analysis focuses on quantifying benefits of improving access to rare disease at an aggregate level. It does not comment on the costs of achieving these benefits, nor individual cost-effectiveness levels which would vary by specific medicines and the policy levers used to achieve them.
Better treatment access also has the potential to improve the efficiency of the health and social care system – slowing disease progression, stabilising rare diseases, and reducing the need for resource intensive care.
“Because of new disease modifying treatments, for a lot of patients, their spinal muscular atrophy has stabilised. This means that a lot of these primary and secondary care visits can be avoided – this can save NHS a lot of resources.” (Charlotte Roy, Muscular Dystrophy UK)
“If you can keep people in earlier stages of motor neurone disease for longer, you can reduce costs associated with long-term ventilation or tracheostomy - then obviously there’s going to be a huge health economic benefit in addition to improvement in quality of life.” (Brian Dickie, MND Association)
1 Introduction
1.1 The case for investing in rare diseases
A rare disease is defined as a condition that affects fewer than one in 2,000 people. While individually rare, when taken together rare diseases affect over 3.5 million people in the UK. Although progress has been made in the diagnosis and treatment of rare diseases, these conditions still have a significant impact on life expectancy and quality of life.9
The impact is particularly significant for children, as they make up a significant proportion of all rare disease cases, with estimates ranging from 25 per cent10 to over 50 per cent.11 Over 30 per cent of children with a rare disease die before their fifth birthday.12
People living with a rare disease often struggle to access the healthcare they need due to three challenges:
- The awareness challenge. There are between 6,000 and 8,000 individual rare diseases. This means that, while individually rare, together there are over 3.5 million people in the UK living with a rare disease.13 Raising awareness of rare diseases is a key step in improving access to care and resources.
- The diagnosis challenge. On average people have to wait 5.6 years after onset of symptoms to receive a rare disease diagnosis.14 This delays the ability of people to access care and support for their condition, and the diagnosis process itself can often have a significant impact on a person's quality of life.
- The evaluation and access to medicines challenge. Only 5 per cent of rare diseases have an effective treatment.15 Even where these do exist, fewer than 35 per cent of treatments licensed in the EU are fully accessible (inclusion on the public reimbursement list) to patients in England and Scotland, and similar levels for NI and Wales who typically adopt NICE recommendations.16 Taking into account medicines licensed elsewhere but not in the EU, this percentage for full access in England and Scotland would be even lower; for example, evidence on FDA and EMA orphan medicine approvals between 2000 and 2022 found that just under 30 per cent of orphan medicines approved by the FDA were either not listed by the EMA or refused EMA authorisation.17 18 This means that there is significant unmet need amongst rare disease patients even after receiving a diagnosis.
These challenges have clear personal impacts for people living with a rare disease, as well as for their loved ones and carers. They also have a wider societal cost to the economic development of the UK. People living with a rare disease are almost three times more likely to be unemployed due to their condition compared to people without disabilities.19 Those in work have more sick days and face limitations in the jobs they can take and barriers to promotion.20 In addition, half of new rare disease cases are in children, and with the burden of care upon parents, these often take time off work or reduce their working hours.21
There is widespread recognition of the challenges facing people living with rare disease, and a shared ambition across government and the NHS for ongoing improvement. The UK Rare Diseases Framework22 sets out four key priorities for improving care for people with rare disease:
- Helping patients get a final diagnosis faster;
- Increasing awareness of rare disease amongst healthcare professionals;
- Better coordination of care; and
- Improving access to specialist care, treatments and drugs.
Box 1: Definitions used in this report
Rare disease is defined in the UK as conditions that affect fewer than 1 in 2,000 people.
Orphan medicines refer to medicines used to diagnose, treat or prevent a rare disease.
Disease modifying treatments target the underlying cause of a disease rather than managing symptoms.
Treatment availability refers to the existence of a licensed treatment.
Treatment access refers to whether a licensed treatment is available and reimbursed on the NHS to all patients.
1.2 Purpose of this report
Improving access to specialist care, treatments and medicines is one priority of the UK Rare Diseases Framework. This study contributes to the evidence base on the value that would be generated by improving access to treatment for people living with a rare disease.
The study takes a societal perspective of benefits to individuals, the health system and productivity. In doing so, it recognises the role that improving treatment access could play in contributing to government objectives of economic growth and supporting the NHS.
The study focuses on benefits associated with improving treatment access. It does not estimate the costs associated with increasing treatment access. The study includes rare cancers under the umbrella of rare diseases.
1.3 Acknowledgements
The authors are grateful to stakeholders who were interviewed to provide data and information for this study.
Interviews were conducted between January and February 2026 with Charlotte Roy, Life Sciences Policy and Influencing Manager at Muscular Dystrophy UK, the leading charity for people in the UK living with muscle wasting conditions; Brian Dickie and Alex Massey, respectively Chief Scientist and Head of Campaigning at the MND Association, a membership organisation dedicated to improving the lives of those affected by Motor Neurone Disease; and Nick Meade, Chief Executive at Genetic Alliance UK, an alliance of over 220 charities and support groups working together to improve the lives of people in the UK with genetic, rare and undiagnosed conditions.
Further interviews were conducted in February 2026 with healthcare providers involved in the care of spinal muscular atrophy (SMA) and with motor neurone disease (MND) patients. We thank Lisa Edel, Senior Specialist Respiratory Neuromuscular Physiotherapist from Great Ormond Street Hospital for Children; Professor Nigel Leigh from the Brighton and Sussex Medical School at Universities of Brighton and Sussex; Dr Kelechi Ugonna, Respiratory and Sleep Consultant, from Sheffield's Children Hospital; and Kay White, Paediatric Neuromuscular Physiotherapist, from Sheffield's Children Hospital.
1.4 Funding declaration
This report was commissioned by the Association of the British Pharmaceutical Industry (ABPI).
Box 2: Approval process for new medicines
New medicines must undergo two separate approval processes before they are available to patients on the NHS.
The UK medicines regulator (the Medicines and Healthcare products Regulatory Agency (MHRA)) assesses its quality, safety and efficacy to determine whether the medicine should be licensed for use. This allows it to be sold and supplied in the UK.
Separately, the medicine must undergo a cost-effectiveness assessment to determine whether it can be provided on the NHS – this is often referred to as reimbursement. This cost-effectiveness assessment is conducted by the National Institute for Health and Care Excellence (NICE) for England, Wales, and Northern Ireland, and by the Scottish Medicines Consortium (SMC) for Scotland. In addition, the All Wales Medicine Strategy Group can conduct its own assessment of new medicines that do not have a NICE assessment.
A treatment must pass both approval processes to be fully accessible to patients on the NHS.
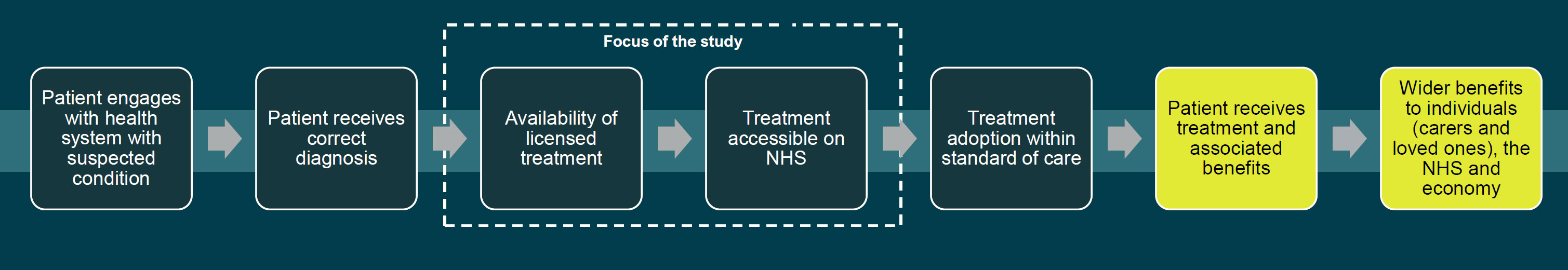
2 Overview of approach
The process of any given person receiving a treatment reflects the combined effect of several factors. They must first receive a diagnosis, which is often a challenging and time-consuming process for rare disease patients. Once diagnosed, the question then turns to whether a licensed treatment is available (discovery and licensing of new treatments) and, if so, whether it is accessible to patients on the NHS (treatment reimbursement). Finally, speed and equity of adoption will determine whether patients can access new medicines once approved for use on the NHS.
This study focuses on the benefits to individuals, the health system and UK economic growth from improving treatment availability and access.
The analysis adopts a mixed methods approach. It combines top-down analysis on the average impact of improvements in treatment availability and access for rare diseases with qualitative indication-level deep-dives. The scope of top-down analysis includes rare cancers; the results excluding rare cancers can be found in Annex B and Annex C.
2.1 Approach to top-down analysis
The top-down analysis models the impact of improving treatment availability and access based on the current R&D pipeline and the UK's Life Sciences Sector Plan ambitions. It defines three scenarios which vary across the proportion of treatments which are accessible on the NHS:
- Base case: This reflects the current clinical and policy environment.
- Enhanced access scenario: This scenario reflects two advances relative to the base case: (1) an expansion in the availability of licensed treatment, informed by the current orphan medicines pipeline, and (2) improved treatment access via NHS reimbursement, modelled as the UK reaching the top three European countries for access to licensed orphan medicines, in line with UK Life Sciences Sector Plan ambitions to be amongst the top three countries in Europe for speed of patient access to medicines.
- Best in Europe access scenario: Compared to the enhanced scenario, this scenario models the impact of the UK becoming the best country in Europe for access to orphan medicines on the NHS.
2.2 Approach to the indication deep dives
The top-down analysis has been complemented by three indication-specific deep-dives:
- Spinal muscular atrophy (SMA)
- Neuromyelitis optica spectrum disorder (NMOSD)
- Motor neurone disease (MND)
These case studies draw together evidence from the literature and stakeholder interviews across patient charities and healthcare providers to understand how treatment affects patients, families and the NHS in practice. The deep dive areas were selected to reflect a diverse range of rare diseases in terms of disease area, age of onset and treatment availability, while ensuring sufficient supporting evidence in the wider literature and avoiding duplication of existing evidence.
Box 3: Basis for scenarios
| Scenario | % of patients for whom a licensed treatment is available | per cent of treatments fully accessible on the NHS |
|---|---|---|
| Base | 5% | 35% |
| Enhanced access | 13% | 50% |
| Best in Europe access | 13% | 89% |
Base case
- Treatment availability. Currently only around 5 per cent of rare diseases have an effective treatment.23
- Treatment access in the UK. Research has found that of those orphan medicines licensed by the European Medicines Agency (EMA), only 38 per cent and 32 per cent are fully available (inclusion on the public reimbursement list) to patients on the NHS in England and Scotland respectively.24 Given NICE guidance is reflected in Wales and Northern Ireland, we take the average of these as a proxy of average orphan medicine reimbursement rates in the UK.
Enhanced access scenario
- Treatment availability. Increase in the proportion of patients who have a treatable rare disease from 5 per cent to 13 per cent based on evidence on the orphan medicine pipeline. An analysis of US Food and Drug Administration (FDA) data found that up to 11–15 per cent of rare diseases have at least one medicine that has been developed and shown promise in their treatment, diagnosis or prevention.25 We take the average of this range to model the potential increase in treatment availability.
- Treatment access in the UK. The UK Life Sciences Sector Plan sets out an ambition that, by 2030, the UK will be one of the top three fastest places in Europe for patient access to medicines and MedTech. Looking across 2020–2023, the third highest full availability rate for orphan medicines in Europe (measured as a per cent of all EMA licensed products) was 50 per cent, which forms the basis of this scenario.
Best in Europe access scenario
- Treatment availability. Same assumptions as in the enhanced access scenario.
- Treatment access in the UK. Across 2020–2023, Germany had the highest full availability rate for orphan medicines at 89 per cent, which forms the basis of this scenario.
Further details can be found in Annex A
3 Value to patients
Access to a treatment can make the difference between a managed and stable condition and rapid decline. Many treatments deliver transformative benefits for patients by stabilising disease, slowing progression and improving quality of life, even when they are not curative.
When licensed treatments are reimbursed and accessible on the NHS, the difference it can make to health outcomes compared to no treatment is often profound. It may lead to slower progression of disease, reduced burden of symptoms and an overall increase in health, quality of life and life expectancy.26 Looking forward, recent medical innovations such as advances in gene therapy provide hope that the future of rare disease treatment can shift from lifelong disease management to disease modifying treatments.
However, as of January 2025, only 38 per cent of all EU approved orphan medicines were fully available to patients in England (with this figure used as a proxy for Wales and Northern Ireland given NICE guidance also applies there).i ii In Scotland this percentage was 32 per cent.29 Even after including medicines made available to restricted cohorts,iii around half of EU approved orphan medicines have no availability in England and Scotland.
Access to effective treatment may not only transform the lives of patients, but can also deliver benefits that extend to carers and wider family members. Evidence shows that unmanaged rare disease is associated with substantial and sustained reductions in quality of life for families, reflecting the emotional impact of living with a progressive or unstable condition and the day-to-day burden of ongoing care.30
Recent health economics research demonstrates a positive relationship between patient health and carers' health-related quality of life.31 When treatments are available and accessible, the resulting potential for stabilisation of disease, slowing of progression and improvement in symptoms can lead to meaningful improvements in family wellbeing.
95% of rare diseases have no licensed treatment27
Over half of the rare disease treatments licensed in the EU are not available to patients in the UK (including full or restricted access)28
“I think it's exciting times, particularly in the ALS field [Amyotrophic lateral sclerosis]. I think it's one of the most dynamic areas in terms of neurology and disease modifying therapies.”
Professor Nigel Leigh, Brighton and Sussex Medical School
“There's a lot of children with SMA that maybe wouldn't have been here if not for the genetic therapy and who have a visibly better quality of life. [...] We are having to transition a lot more patients into adulthood.”
Dr Kelechi Ugonna, Sheffield's Children Hospital
“We'd like patients to have a more equitable experience at the point of diagnosis – for example, make sure you get a message from your clinician with information about your rare condition, the treatments available, the existing research and the community that you are now part of.”
Nick Meade, Genetic Alliance UK
In addition to economic benefits, improving treatment accessibility can contribute to improving equity among patients living with rare diseases. Genetic Alliance UK, an alliance of over 200 charities and support groups, focussed on equity for their Rare Disease Day 2026 campaign, advocating for patients' right to fair and equitable care, regardless of how rare their disease. Addressing equity concerns does not only imply an effort to provide equitable access to treatments over different areas in the country and across different conditions, but also fighting for more equity in research and in experience at point of diagnosis.
Using the modelled scenarios, we quantify the lifetime impact of improving treatment access through NHS reimbursement, alongside an expansion in the number of rare diseases with a licensed treatment on health outcomes for people living with a rare disease.
Under the enhanced access scenario, the increase in treatment access from better reimbursement rates on the NHS combined with the expected expansion in licensed treatment availability leads to £2.1 billion of additional QALY benefits compared to the base case. This increases to £4.4 billion under the Best in Europe access scenario, which has a higher reimbursement rate.
We model an additional scenario in which NHS access improves while treatment availability remains at current levels (i.e. 5 per cent of rare diseases have a licensed treatment without assuming expansion from the pipeline).
- Under the assumption that the UK moves to the top three best countries for treatment access, improving treatment access via NHS reimbursement alone leads to additional QALY benefits of £0.4 billion.
- Assuming, instead, that the UK becomes the best in Europe for treatment access, additional lifetime QALY benefits amount to £1.3 billion.
To realise the full potential of the orphan medicine pipeline, improvements in NHS access are essential. An expansion in licensed treatment options increases the number of patients who could benefit, but the scale of realised benefit depends on the proportion of those treatments that are accessible on the NHS.
The UK becomes the third best place in Europe for access to orphan medicines
The UK becomes the best place in Europe for access to orphan medicines

76,000 more patients receiving treatment
158,000 more patients receiving treatment

33,000 gain in lifetime QALYs (discounted), valued at £2.1 billion
68,000 gain in lifetime QALYs (discounted), valued at £4.4 billion
These estimates are likely to be conservative for three reasons:
- Assumptions on the diagnostic odyssey. The analysis is based on the current experience of rare patients in receiving a final diagnosis. However, diagnosis is a major challenge for accessing treatment, with the average patient waiting 5.6 years from the onset of symptoms before receiving a diagnosis.32 The NHS 10 Year Plan sets out an ambitious commitment to improving the diagnosis of rare disease.33 If achieved, this would increase the number of patients able to access treatments, thereby amplifying the benefits estimated under the same modelling assumptions.
- Available data on prevalence. Diagnosed prevalence of rare diseases in the UK is estimated at 1.5 per cent, compared with a global point prevalence of approximately 3.5 per cent reported in Orphanet.34 This gap is partly driven by the diagnostic odyssey described above and partly by limitations in NHS electronic health record coverage and clinical coding, which constrain the identification of a large share of rare diseases. As a result, prevalence estimates based on diagnosed patients are likely to understate the true population affected.
- QALY benefit of new treatments. The QALY gains associated with treatment discovery are based on backward-looking evidence from orphan medicine approvals between 1999 and 2019. Over this period, the median incremental lifetime QALY gain reported for orphan medicines is 0.43, which is the value used in this analysis. However, if we were to take the mean QALY estimate rather than the median from this same review, this would give us an incremental lifetime QALY gain of 0.99 (further details in Annex E). The mean QALY estimate is higher than the median as it reflects the small number of breakthrough treatments to date that can generate significant QALY gains. As we see more developments in areas such as gene therapy and other advanced therapies that could deliver potentially curative treatments for rare diseases, it is possible that future innovations will deliver larger average health gains than those observed historically so the mean QALY estimate may be a reasonable forward-looking perspective.
SPINAL MUSCULAR ATROPHY (SMA)
SMA is a rare disease that causes muscle weakness, leading to significant disability, paralysis and often death. The majority of patients suffer from Type 1 SMA with symptoms developing between two and six months of age, and the historically poor prognosis means the majority of children are not expected to survive beyond the age of two without respiratory ventilation. Around 70 children are born with SMA each year in the UK and it is the most common genetic cause of infant mortality. Over the last decade, however, there have been major advances in treatment for SMA with the development of disease modifying therapies (DMTs), which have drastically changed the standard of care for patients.
Step change in treatment options
"One of the greatest benefits of these new treatments is that they can stabilise the disease and stop symptoms from worsening. This has an impact not only on patients, who now have a better chance of transitioning into adulthood, completing education and starting work, but also on carers, easing burden and allowing them to re-enter the workforce. It can also result in needing less specialist support and equipment." (Charlotte Roy, Muscular Dystrophy UK)
"SMA kids that receive treatments have got increased function, better head control, so they're tolerating being sat up or being positioned in sitting. They've got less fatigue, they've got some upper limb function, they can hold things, they can move their arms a little bit, even if they can't raise their arms above their head, they can hold a lightweight toy. It's been quite small improvements in motor function, but I think those small movements, and those small abilities have massively impacted on quality of life." (Kay White, Sheffield's Children Hospital)
"The need for respiratory support has broadly gone down. We're having to start only a few patients on ventilation and if we are starting, we're starting later. An SMA patient who didn't need ventilation was unheard of and we have a few now, two or three or four year olds who are managing without the ventilators and just use cough assist." (Dr Kelechi Ugonna, Sheffield's Children Hospital)
These new treatments have been transformative for children with Type 1 SMA, more than halving the number of deaths per year and increasing the proportion of children who reach the age of two to almost three quarters.35
Currently, three main treatments are being offered in the UK, although the conditions for access can vary.
Varied access for existing treatment
"There are three treatments that are available at the moment for people with SMA. One that has been fully approved (a gene therapy) which delivers the functional version of the gene - this is only available for children under two under a certain weight limit. The other two target SMN2 to deliver the backup protein that is missing - one is a solution/tablet, and one is intrathecal injection four times a year. These are on managed access, and the provision of one or the other depends greatly on patients' conditions and local health system capacity." (Charlotte Roy, Muscular Dystrophy UK)
In addition to advances in treatment options, the early onset and rapid decline associated with untreated SMA means that early diagnosis is a key step in improving patient outcomes. The UK Newborn Screening Committee published a progress blog which included a breakdown of evidence for SMA screening, and all the analyses have supported the view that screening could result in better outcomes and lower costs compared to the current approach of no screening plus treatment.36
In addition to the patient impact, the early onset nature of SMA means that the burden of care often falls on patient families. Caregivers for people with a rare disease provide on average 10 hours of care a day, and work impacts are common. Of those caregivers that are employed, over half experience work related problems due to caregiving, 30 per cent take days off work, and over a third experience difficulties meeting work schedules.37 Oftentimes, caregivers report needing to stop work altogether.38 The advance of new treatments has helped to alleviate the need for this in some cases.
Importance of meeting treatment window
"With treatments delivered early on, babies are developing as expected and reaching key milestones, which in turn has a massive impact on the kind of healthcare resources they need further down the line. This is why newborn screening is so crucial and we have been working for many years now to try and get this through the newborn screening committee." (Charlotte Roy, Muscular Dystrophy UK)
"The fact that we don't have newborn screening in this country has a huge impact on NHS resource utilisation. If we had newborn screening and we were treating these kids pre-symptomatically, yes, for sure we would have less utilisation of NHS resources. However, we are still treating these patients once they are symptomatic and therefore the utilisation on resources is still massive." (Lisa Edel, Great Ormond Street Hospital for Children)
Impact on carers
"Parents are often the primary caregivers, and these treatments have had a huge impact on them. From the physical burden to the emotional aspect of not dreading the progress of the disease - everything has been impacted. And now more children transition into adulthood as well, which means that the caregiving responsibilities can shift from parents to social care or paid caregivers. A lot of parents can actually go back to work." (Charlotte Roy, Muscular Dystrophy UK)
4 Value to economic growth
The impact of living with a rare disease, or caring for a loved one with a rare disease, often limits one's ability to work. Improving treatment access helps to lessen this burden and has knock-on impacts on economic growth.
Rare diseases not only have a significant impact on an individual's wellbeing but they also present an economic cost to the nation. Working-age people living with rare diseases are almost three times as likely to be unemployed than the average population, while those in work report high levels of absenteeism and lower productivity.39
Furthermore, children make up a significant proportion of all rare disease cases, with estimates ranging from 25 per cent40 to over 50 per cent.41 The responsibility for providing care for these children often falls on one or both of their parents, resulting in parents reducing their hours of employment or leaving the workforce altogether. While current treatments for rare diseases cannot fully alleviate this burden, treatment is associated with a significant reduction in the productivity loss associated with rare disease.42
A European survey of 3,000 rare disease patients and carers found that:43

70% of patients and carers had to stop or reduce professional activity due to rare disease44

69% of patients and carers experienced an income decrease due to rare disease

Delayed diagnosis and limited treatment options for rare disease costs the economy £14.9 billion a year45
We estimate that:
- Under the Enhanced access scenario, with access reimbursement rates in the top three in Europe, annual productivity benefits increase to approximately £1.1 billion annually (discounted).
- Moving to the more ambitious, Best in Europe access scenario, would deliver £2.3 billion of productivity benefits (discounted).
- Under this scenario, improving access through reimbursement alone, with treatment availability unchanged, would generate £0.8 billion a year in additional GVA, driven by improved labour market participation among working-age patients and carers of children with a rare disease.
As with benefits to health outcomes, availability and access reinforce one another. To realise the full potential of the orphan medicine pipeline, improvements in NHS access are essential. An expansion in licensed treatment options increases the number of patients who could benefit, but the scale of realised benefit depends on the proportion of those treatments that are accessible on the NHS.
£19.4 BILLION IN CUMULATIVE PRODUCTIVITY BENEFITS OVER 10 YEARS

Under the Best in Europe access scenario, we estimate £19.4 billion in discounted productivity benefits over a ten-year period. We present these cumulative benefits assuming that effective intervention occurs at least ten years before retirement for the typical patient or carer. This assumption is grounded in UK NHS data which shows that the average age of the working-age rare disease population is 41. We do not adjust GVA with any expectation of future growth and roll forward annual benefits over ten years from current steady-state estimates.
In addition to productivity benefits from a healthier and more productive population, we also estimate the impact on avoided Carer's Allowance payments under the Best in Europe access scenario. While this would represent a transfer between individuals (from those currently in receipt of the Carer's Allowance to government and subsequently taxpayers), it is a useful illustration of distributional impacts.
£100 MILLION IN AVOIDED CARER'S ALLOWANCE PAYMENTS ANNUALLY

These savings are estimated by inferring the number of carers who would return to employment if their child's rare disease were effectively managed, consistent with the approach used to estimate productivity benefits. Estimates assume one primary carer per child and are valued using the statutory Allowance rate. Savings are reported separately from productivity impacts as Carer's Allowance represents a fiscal transfer between government and individuals. They represent a direct, cash-releasing benefit to government.
More broadly, evidence on industry clinical trials demonstrates that strengthening the UK's access and innovation environment can support substantial wider economic activity. In 2022 alone, industry clinical trials contributed £7.4 billion in GVA and supported 65,000 jobs across the UK economy, including 13,000 roles within the NHS. Previous analysis suggests that restoring trial activity to peak levels could generate a further £3 billion in GVA and 26,000 additional jobs. These benefits relate to innovative medicines generally and highlight the wider growth potential associated with a strong R&D ecosystem.51
There are several reasons why this is likely to be a conservative estimate of annual productivity impacts. First, it assumes no change in the current speed and accuracy of diagnosis. In reality, the significant investments in early diagnosis will amplify these benefits by identifying a greater number of patients who are able to benefit from treatments.
Second, a substantial share of the rare disease population are children. Children with a rare disease are more likely to experience disruption to education, driven by frequent healthcare utilisation, hospital admissions and ongoing clinical management, which can limit regular school attendance and participation, and future career opportunities.52 Survey evidence indicates that the majority of pupils with a rare disease experience restricted participation in education,53 while UK cohort evidence demonstrates significantly higher risks of underachievement in early educational milestones compared with peers without rare disease.54
Where the development and access to new treatments can deliver significant improvements in health outcomes, and this translates into improved educational attainment, this could have longer-term economic consequences that have not been captured in this analysis. Finally, this analysis does not quantify the impact of avoided premature mortality. Other studies have found that this is a significant contributor to lost productivity due to untreated rare disease, particularly given the early onset nature of many rare diseases.55
MOTOR NEURONE DISEASE (MND)
MND is an umbrella term for a group of progressive, terminal neurodegenerative disorders that cause muscle weakness, loss of mobility and increasing difficulty with speech, swallowing and breathing, with death usually resulting from respiratory failure. Most people diagnosed with MND have the "mixed" form affecting both upper and lower motor neurones (often referred to as amyotrophic lateral sclerosis – ALS), while a smaller minority have rarer phenotypes such as progressive muscular atrophy (PMA) (predominantly lower motor neurone involvement) or primary lateral sclerosis (PLS) (predominantly upper motor neurone involvement). It typically begins in later working life, commonly between ages 50 and 70, but adults of any age can be affected.
Heterogeneity in MND
"As clinicians and researchers, we have a big problem with the heterogeneity in MND – this is a syndrome, not one disease. [...] We have known for many years that there are different clinical types of MND, often overlapping, and providing a different set of challenges for day-to-day care." (Professor Nigel Leigh, Brighton and Sussex Medical School)
The disease course is often short and severe. The Demos "Financial Impact of MND" report notes average life expectancy of three to five years from symptom onset, with around one-third dying within a year of diagnosis and more than half within two years. As for other neurodegenerative diseases, and even more so due to the short life-span of patients after onset, early diagnosis is key.
Diagnostic odyssey
"You need rapid referral to a neurologist. And at the moment, in the UK, too often that is not happening. There may be months of delay and people are increasingly having to go privately to get round that obstruction." (Professor Nigel Leigh, Brighton and Sussex Medical School)
"Currently the average time for a MND diagnosis is around 12 months. If you can keep people in those earlier stages for longer, you can reduce costs associated with long-term ventilation or a tracheostomy - then obviously there's going to be a huge health economic benefit in addition to improvement in quality of life." (Brian Dickie, MND Association)
The current approach to treatment, although mainly palliative, has greatly improved the conditions of MND patients. The focus is on coordinated multidisciplinary care, with NICE guideline NG42 setting out the role of specialist MND multidisciplinary teams and the MND Association highlighting improvements in quality of life and reduced unplanned admissions.
Importance of coordinated care
"The thing that has transformed MND since I started in the field is the coordinated multi-professional approach to care. It wasn't there before and it's beyond anything else that I've seen change for the better [...] There is now evidence showing that coordinated care does keep people out of hospital. And instead of going into hospital for acute care in a crisis, people can access palliative care at home or in a hospice. They don't need to use the acute hospital system as much." (Professor Nigel Leigh, Brighton and Sussex Medical School)
"There are three key components to coordinated care that may impact survival and quality of life. The first is access to a disease modifying drug, which, if well tolerated, can have a significant impact on survival especially for people in the early stages of the disease. The second is access to non-invasive ventilation, which improves survival and quality of life and may reduce respiratory crises that drive emergency admissions and intensive care use. The last is nutritional management, as MND patients may have higher metabolisms and burn energy at a slightly faster rate than the general population. It is important they maintain a good energy intake when diagnosed, because motor neurons are the most energy hungry cells in the body." (Brian Dickie, MND Association)
Current research is increasingly targeting biologically defined (genetic) subtypes, which are individually rare even within the MND/ALS population. As MND is increasingly segmented into smaller clinical and genetic subtypes, discussions on further R&D investments should consider trade-off between spreading limited resources across a larger number of potential treatments (each applicable to a smaller patient group) versus focusing resources on a smaller number of programmes with broader applicability.
Advances in treatment
"The effect of this new treatment on individuals with the SOD1 mutation can be significant. We have heard of people whose disease has plateaued, and others for whom some function has been restored. This is why we have actively campaigned for NICE to conduct a highly specialised appraisal of the drug, which is scheduled to progress in the next few months." (Brian Dickie, MND Association)
6 The case for change
Rare diseases collectively affect millions of people in the UK, yet for most conditions effective treatment remains unavailable or difficult to access. The government has set out a mission to address this challenge, including objectives to improve treatment availability in the UK Rare Diseases Framework and 10 Year Plan.
This analysis demonstrates the value of delivering against these objectives by improving access to innovative rare disease treatments and ensuring that the NHS is positioned to adopt medicines emerging from the current development pipeline.
Under the Best in Europe access scenario, where the UK matches the highest observed European access reimbursement performance and the percentage of rare diseases with an effective treatment increases to 13 per cent consistent with pipeline estimates, this could deliver significant benefits to patients, the economy, and the NHS.

Benefits to patients
158,000 more patients receiving treatment delivering 68,000 additional lifetime QALYs (discounted), valued at £4.4 billion.

Benefits to the economy
£2.3 billion of annual productivity benefits from reduced absenteeism and increased workforce participation, equivalent to £19 billion over 10 years.

Benefits to the NHS
Better treatment access also has the potential to improve the efficiency of the health and social care system – slowing disease progression, stabilising conditions, and reducing the need for intensive and resource intensive care.
Figures refer to the Best in Europe access scenario
Improving access to innovative rare disease treatments via NHS reimbursement may also help reduce pressures elsewhere in the system. Better disease management can lessen reliance on acute care and social care services. Where treatment reduces caregiving burden and enables carers to return to work, there are fiscal implications: avoided Carer's Allowance payments are estimated at approximately £100m per year under the Best in Europe access scenario.
Against this backdrop, the analysis in this report shows that the annual productivity benefits alone associated with improved access are substantial relative to current levels of spending on rare disease treatments, even before accounting for the health gains experienced by patients. These benefits accrue beyond the medicines budget, across households, the labour market and public services, and over time.
The modelling presented in this report provides a steady-state estimate of the health and productivity gains associated with improved access. Further work could deepen understanding of timing and system impacts, including dynamic modelling of patient flows, more granular disease-level analysis and integration of improvements across the full patient pathway such as faster diagnosis.
However, the evidence is clear: improving access to innovative rare disease treatments delivers meaningful benefits for patients, carers, the NHS and the wider economy. Realising the full value of the existing treatment pipeline requires that scientific progress is matched by effective NHS access. Doing so would not only improve outcomes for people living with rare diseases but would also support the UK's broader health and economic growth ambitions.
Annex A Methodology: Description of scenarios
This analysis models a number of scenarios. These scenarios are not forecasts; they are designed to illustrate the scale of benefits that could be realised if key constraints in the rare disease pathway were addressed.
The scenarios focus on two levers in the patient pathway where policy and system performance can materially change outcomes:
- Treatment availability: whether a licensed treatment exists for a given rare disease; and
- Treatment access: whether licensed treatments are fully reimbursed and routinely available to patients through the NHS.
We model three scenarios:
- Base case: This reflects the current clinical and policy environment.
- Enhanced access scenario: This scenario reflects two advances relative to the base case: (1) an expansion in the number of rare diseases with a licensed treatment, informed by the current orphan medicines pipeline, and (2) the UK achieving its Life Sciences Sector Plan ambitions to be amongst the top three countries in Europe for speed of patient access to medicines.
- Best in Europe access scenario: Compared to the enhanced scenario, this scenario models the impact of the UK becoming the best country in Europe for accessibility of orphan medicines on the NHS.
To isolate the contribution of each lever, we model the impact of changing treatment availability and treatment access via reimbursement in isolation for each scenario, and then model the combined impact.
Treatment availability benchmarks
Treatment availability is proxied using the proportion of rare diseases for which a licensed treatment exists. In the base case, this is assumed to be 5 per cent, reflecting widely cited evidence that only a small share of rare diseases currently have an effective treatment.70
In the conservative and ambitious scenarios, this increases to 13 per cent of rare diseases, based on published academic evidence on the orphan medicine development pipeline and the proportion of designated orphan products expected to progress to approval over time.71 This assumption reflects the potential for the current pipeline to be brought to fruition in a supportive research and development environment, rather than assuming a structural change in success rates or clinical effectiveness.
This benchmark is therefore grounded in observed pipeline data and regulatory outcomes and is intended to represent a plausible upper-bound scenario for treatment discovery over the medium term. Given that the future pipeline will not be brought through immediately, the benefits associated with greater access to future treatments are discounted in our model.
Treatment access benchmarks
Treatment access via reimbursement is measured using the EFPIA Patients W.A.I.T. Indicator definition of "full availability" for orphan medicines. We use these indicators to benchmark the UK against observed performance in other European systems. This provides an evidence-based way to define plausible access improvements grounded in published data, rather than selecting arbitrary access rates.
To reflect varying degrees of ambition, we model a staged improvement in access, moving from:
- Base case: 35 per cent based on an average of England and Scotland full availability rate for orphan medicines, and assuming the figure for England is reflected in Wales and Northern Ireland;
- Enhanced access scenario: Top three in Europe (benchmarking to the third highest-performing country, Austria, 50 per cent full availability); and
- Best in Europe access scenario: Best in Europe (benchmarking to the highest-performing country, Germany, 89 per cent).
These benchmarks allow results to be interpreted as step changes associated with reaching progressively stronger access positions, with Best in Europe access representing an upper-bound scenario and the Enhanced access benchmark providing a minimum ambition consistent with the Life Sciences Sector Plan ambitions.
What changes across scenarios
To model these scenarios in a transparent way, we change only two parameters relative to the base case:
- Treatment availability (the proportion of rare diseases with a licensed treatment); and/or
- Treatment access (the proportion of licensed treatments that are fully available on the NHS).
All other aspects of the model, including population size and diagnosis rates, per patient health gains, productivity parameters and valuation assumptions, are held constant across scenarios. The scenarios therefore operate by changing the number of people with a rare disease who receive effective treatment.
Annex B Methodology: Value to patients
This annex sets out the approach used to estimate the health benefits under each scenario. The analysis focuses on incremental health gains, expressed in quality-adjusted life years (QALYs), and values these gains using alternative QALY valuation benchmarks.
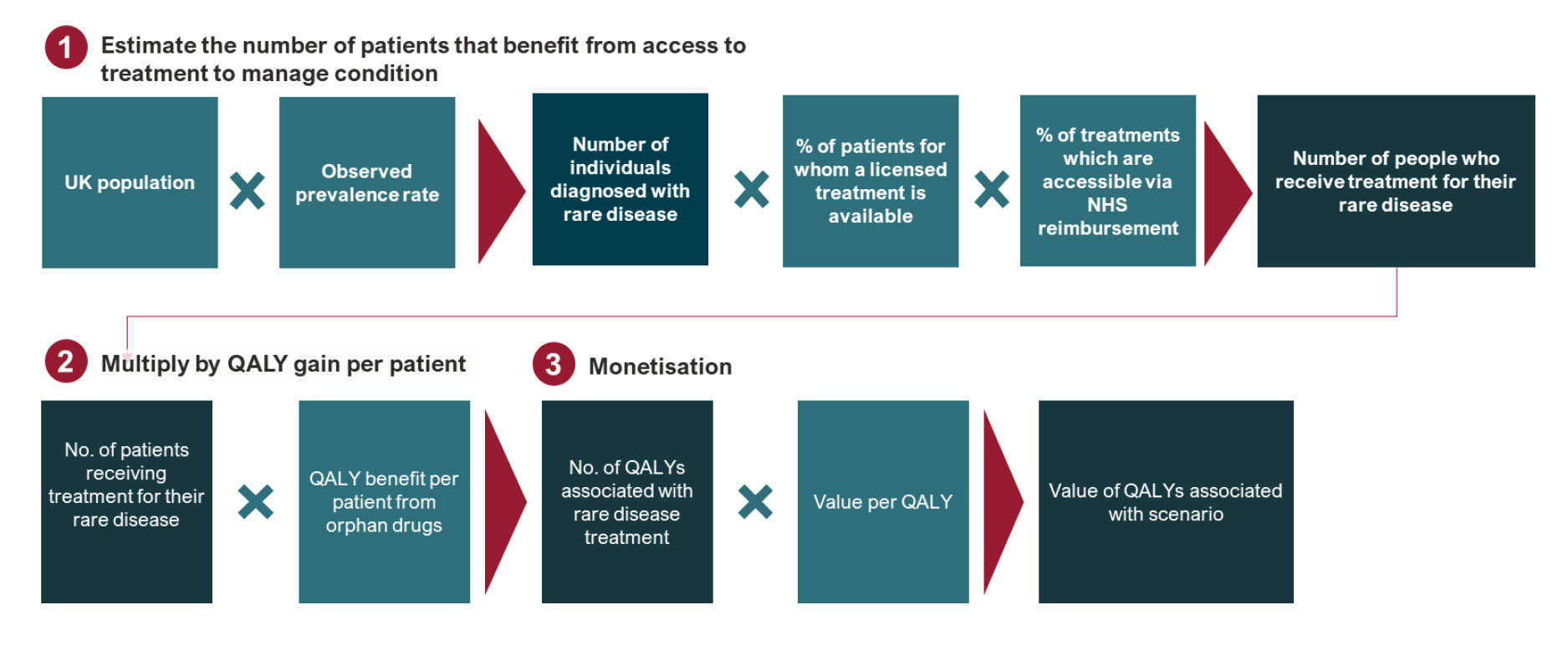
The patient benefits analysis follows four steps. (See figure below for high level summary of steps 1–3):
- Estimate the number of patients receiving treatment under each scenario and base case;
- Apply evidence-based incremental QALY gains per treated patient;
- Value incremental QALYs using Treasury Green Book QALY values; and
- Apply discounting to reflect the timing of treatment availability for benefits associated with treatment discovery.
Step 1: Estimating the treated patient population
The analysis begins by estimating the number of patients receiving treatment for their rare disease under each scenario. This is derived from:
- The size of the rare disease population;
- The proportion of patients diagnosed;
- The proportion of rare diseases for which a treatment exists (treatment availability); and
- The proportion of eligible patients who are able to access treatment via NHS reimbursement (treatment access).
The base case access rate reflects current UK performance and is calculated as the average of access levels observed in England and Scotland (assuming that the figure for England is reflected in Wales and Northern Ireland), providing a representative national benchmark. Alternative scenarios increase access reimbursement rates.
The estimation of the treated patient population draws on observed prevalence estimates from large-scale, NHS-linked electronic health record (EHR) data.72 These estimates provide an important and consistent basis for modelling but should be interpreted as conservative lower-bound estimates of the true rare disease population. Evidence from a recent population-wide study using linked EHR data for more than 58 million people in England illustrates why observed prevalence is likely to underestimate true prevalence for many rare diseases. That study reported point prevalence estimates for 331 rare diseases, including new prevalence estimates for 184 conditions not previously characterised in Orphanet. However, these 331 diseases represent only a small fraction of the over 10,500 clinical entities listed in Orphanet.
Additionally, many people with rare diseases remain undiagnosed or misdiagnosed for extended periods. As a result, they do not appear in coded health records under a rare disease diagnosis, even though they experience disease-related morbidity and healthcare utilisation. This means that prevalence based on observed diagnostic codes will exclude a proportion of affected individuals.
For the purposes of this analysis, observed prevalence estimates are therefore used as a pragmatic and transparent starting point, recognising that they are likely to understate the true number of people living with rare disease. This reinforces the conservative nature of the estimates presented and is consistent with the broader evidence on the diagnostic odyssey in rare disease.
The observed prevalence estimates derived from NHS-linked EHR data do not explicitly identify rare cancers within the set of 331 rare diseases reported. Review of the listed conditions suggests that rare cancers are not substantively captured within the baseline prevalence estimate used in this analysis.
To incorporate rare cancer patients, we draw on the NHS Rare Conditions Registration statistics (2022).73 In these statistics, rare cancers are identified separately via the National Cancer Registration Dataset and classified in line with the US National Cancer Institute's Surveillance, Epidemiology and End Results (SEER) definition. This reflects differences in underlying data sources rather than a conceptual distinction in rarity.
We mapped the "Disease/condition grouping level 1" categories in the Rare Conditions Registration dataset to cancer versus non-cancer conditions and compared the reported prevalence of diagnosed rare cancers with other rare diseases. On this basis, rare cancers account for approximately 35 per cent of patients diagnosed with a rare condition in the registration dataset. This implies that the baseline non-cancer rare disease population represents approximately 65 per cent of the total, equivalent to a 55 per cent uplift when rare cancers are included.
This 55 per cent share is applied as a proportional uplift to the rare disease population underpinning the QALY and productivity modelling. The Rare Conditions Registration dataset captures a smaller total patient population than the broader EHR-based prevalence estimate used in the core model. However, it provides the most consistent NHS-published source for estimating the relative share of rare cancers within the diagnosed rare disease population. It is therefore used to inform proportional scaling rather than to redefine overall prevalence.
Treatment availability is proxied using a headline estimate of the proportion of rare diseases for which an effective treatment exists. In the base case, this is assumed to be 5 per cent, reflecting widely cited evidence that only a small share of rare diseases currently have an available treatment. This estimate is applied uniformly across the rare disease population and is not weighted by disease prevalence, meaning that it captures the number of conditions with a treatment rather than the share of patients covered. The alternative scenario assumes an increase in treatment discovery to 13 per cent, based on the best available academic evidence on the orphan medicine pipeline and the proportion of designated products expected to progress to approval over time.
Treatment access via NHS reimbursement is estimated using evidence from the EFPIA Patients W.A.I.T. Indicator, focusing on measures of full public availability of licensed orphan medicines.74 The base case access rate reflects current UK performance and is calculated as the average of access levels observed in England and Scotland, providing a representative national benchmark. The improved access scenario assumes that the UK moves to the best-performing country in Europe, benchmarked against Germany, which currently exhibits the highest levels of access according to the W.A.I.T. indicators. We also show the results if the UK were to move to the third best country in Europe.
The estimates are expressed in steady-state terms, representing the number of patients receiving treatment once the system has fully adjusted to the scenario assumptions. The analysis does not model year-by-year patient flows, uptake curves or transitional dynamics.
Step 2: Incremental QALY gains per treated patient
Health benefits are measured as incremental QALY gains per treated patient, reflecting improvements in survival and health-related quality of life relative to the previous standard of care.
Incremental QALY estimates are sourced from a recent peer-reviewed study that synthesises evidence across orphan and ultraorphan medicines using the Tufts Health Gains in IQ database.75 This study does not model health outcomes directly or apply its own discounting. Instead, it extracts incremental QALY gains exactly as reported in published cost-effectiveness analyses (CEAs) of orphan medicines. As a result, the QALY values used in this analysis are presumed to be already discounted, because the underlying CEAs apply standard discount rates to both costs and health outcomes in line with jurisdictional guidance (for example, NICE, US Panel on Cost-Effectiveness, or other national Health Technology Assessment bodies). The source paper therefore treats these QALY estimates as final discounted outcomes and does not re-profile or re-discount them.
The paper identifies 284 medicines with at least one relevant CEA, of which 73 were approved for at least one orphan disease (defined as prevalence 200,000 to ≥10,000) and 33 were approved for at least one ultraorphan disease (prevalence <10,000).
Table 1 Summary of incremental QALY values
| Outcomes | Orphan medicines | Ultraorphan medicines | Total |
|---|---|---|---|
| Number of medicine-indication pairs | 73 | 33 | 106 |
| Incremental QALYs: Median (IQR) | 0.31 (0.76) | 0.70 (1.43) | 0.43 |
| Incremental QALYs: Mean (SD) | 0.58 (1.13) | 1.89 (5.13) | 0.99 |
Source: Clifford et al, 2024
The paper reports average incremental QALY gains across orphan medicines, pooling results from a large number of CEAs covering different conditions, time horizons and modelling approaches. While there is inevitably some variation in discount rates and time horizons across the underlying studies, this heterogeneity is inherent in the published evidence base and is reflected in the pooled estimates.
For the purposes of this analysis, we apply the average incremental QALY gain per treated patient reported in this paper. It is important to note that applying a single average incremental QALY gain as a one-off per treated patient is a simplifying assumption. While CEAs typically model health benefits over a defined time horizon, treatment effects may persist beyond the period explicitly captured in the underlying models. As a result, this approach is likely to understate the full lifetime health benefits associated with treatment, reinforcing the conservative nature of the patient benefit estimates presented in this analysis.
Step 3: Valuation of QALYs
Incremental QALYs are monetised using alternative values per QALY, reflecting current appraisal practice and uncertainty. The analysis uses a central estimate alongside a sensitivity range:
- Central estimate: £70,000 per QALY (2020/21 prices), based on Green Book supplementary wellbeing guidance. This value reflects a cross-government perspective on the societal value of health improvements.
- Lower bound: £30,000 per QALY, reflecting the midpoint of NICE standard thresholds for non-rare conditions, as per the update planned for April 2026.
- Upper bound: £85,000 per QALY, representing an inflation-adjusted Green Book value (2025/26 prices) to reflect more recent price levels.
Other QALY values considered but not used as central estimates include higher NICE thresholds applied in rare and ultra-rare disease contexts (up to £100,000–£300,000 per QALY).
Step 4: Discounting of patient benefits
- Health benefits associated with treatments that are not yet available to patients are discounted to reflect the time lag between treatment development and realisation of health gains. This is consistent with HM Treasury Green Book guidance, which recommends discounting future health and life outcomes at 1.5 per cent per annum.
- For treatments that are already available, patient health benefits are assumed to accrue immediately and are therefore not discounted. However, for pipeline orphan medicines, health benefits will only be realised once treatments progress through regulatory approval and become accessible to patients in routine care. Discounting is applied to reflect this delay.
The timing of health benefit realisation is based on published evidence on orphan medicine development and access timelines. As the pipeline date is based on an analysis of FDA data, benchmarks on time to licence is based on FDA benchmarks. Time from licence to reimbursement is based on UK evidence.
- The average clinical development duration is 7.2 years.
- Following approval, there is an additional delay before routine patient access. Based on UK evidence on patient access timelines, the average time from approval to access is assumed to be 1.07 years.
Taken together, this implies an average delay of 8.2 years from designation to patient access, at which point health benefits begin to accrue.
These assumptions are based on peer-reviewed regulatory evidence76 and the EFPIA Patient W.A.I.T. Indicator,77 and are intended to represent a central, conservative estimate of the time to real-world benefit.
Discounting is applied using the Green Book health discount rate of 1.5 per cent per year, starting from the point at which health benefits are realised. The cumulative discount factor applied to pipeline treatments reflects the full designation-to-access delay, rather than discounting year-by-year patient flows.
Table 2 Estimated patients benefits including rare cancer (Health-Related Quality of Life value)
| Item | Base case | Enhanced access | Best in Europe access | Source |
|---|---|---|---|---|
| UK population (A) | 69,300,000 | ONS | ||
| Per cent diagnosed with rare disease (B) | 1.50% | Thygesen et al, 2025 | ||
| UK patients diagnosed with rare disease (C) | 1,039,500 | 1,039,500 | 1,039,500 | C = A*B |
| Rare cancer uplift (D) | 55% | Rare condition statistics, NHS | ||
| UK patients diagnosed with rare disease (incl. rare cancer) (E) | 1,611,225 | 1,611,225 | 1,611,225 | E = C*(1+D) |
| Treatment exists to manage condition (F) | 5% | 13% | 13% | UK govt. (base), Fermaglich and Miller, 2023 (scenario) |
| Patient access (% public availability) (G) | 35% | 50% | 89% | Patient W.A.I.T Indicators (base and scenario) |
| Patients with diagnosis and treatment (H) | 28,196 | 104,730 | 186,419 | H = E*F*G |
| QALY benefit with treatment (I) | 0.43 | 0.43 | 0.43 | Clifford et al, 2024 |
| Total QALYs (J) | 12,124 | 45,034 | 80,160 | J = H*I |
| Value per QALY: Green Book appraisal value (in £), unadjusted (K) | £70,000 (£30,000 – £87,500) | |||
| HRQoL value (£billion) (L) | £0.9 (£0.4 – £1.1) | £3.2 (£1.4 – £3.9) | £5.6 (£2.4 – £7.0) | L = J*K |
| Delta HRQoL value (£billion) (M) | £2.3 (£1.0 – £2.9) | £4.8 (£2.0 – £6.0) | M = Lscenario – Lbase | |
| Delta HRQoL value (£billion, discounted) (N) | £2.1 (£0.9 – £2.6) | £4.4 (£1.9 – £5.5) | Green Book discount rate for health and life outcomes | |
Source: Frontier Economics
Note: Estimates shown according to central QALY value of £70,000, analysis was also run using range of £30,000 – £87,500. † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US)
Annex C Methodology: Value to economic growth
Productivity benefits reflect reductions in productivity losses experienced by people living with a rare disease and their informal carers, arising from improved disease management following treatment.
The analysis focuses on avoided productivity losses, valued in monetary terms and expressed as gross value added (GVA) savings. It captures changes in labour market participation and work performance, including absenteeism and reduced productivity while in work.
The productivity analysis follows four steps:
- Estimate productivity loss associated with unmanaged rare disease, for patients and carers;
- Apply evidence-based reductions in productivity loss associated with effective treatment;
- Scale productivity gains by the number of additional patients gaining access to treatment under each scenario; and
- Apply discounting to reflect the timing of benefit realisation and estimate cumulative five- and ten-year productivity benefits.
The analysis is expressed in steady-state terms and estimates the productivity impact once the system has fully adjusted to the scenario assumptions.
Step 1: Productivity loss associated with unmanaged rare disease
The analysis begins by establishing the baseline productivity loss associated with unmanaged rare disease, separately for working-age patients and informal carers of children with rare disease. Productivity losses are estimated in terms of reduced GVA and reflect two distinct channels: reduced workforce participation and absenteeism among those in work.
Working-age patients
Among working-age adults diagnosed with a rare disease, the analysis estimates the number of individuals without access to treatment and therefore living with unmanaged disease. For these individuals, productivity losses arise through both a higher likelihood of being out of work and increased sickness-related absence among those who remain employed.
First, productivity losses associated with reduced workforce participation are estimated by applying evidence on the likelihood of being out of work among people living with a rare disease to the working-age unmanaged population. The resulting number of individuals out of work is multiplied by average output per worker to estimate the total loss of output associated with non-participation. This produces an estimate of lost GVA attributable to reduced workforce participation, expressed both in aggregate and on a per unmanaged patient basis.
Second, productivity losses associated with absenteeism are estimated for working-age individuals with unmanaged rare disease who remain in employment. Absenteeism-related losses are calculated using evidence on the average number of additional days off work attributable to untreated disease, combined with assumptions on hours worked per day and average GVA per hour. This yields an estimate of lost GVA per employed unmanaged patient and total absenteeism-related productivity loss across the working-age population.
Total productivity loss for working-age patients with unmanaged rare disease is calculated as the sum of losses due to reduced workforce participation and absenteeism. This represents the baseline annual productivity loss associated with unmanaged rare disease among working-age adults.
Carers of children with rare disease
Productivity losses are also estimated for informal carers of children living with rare disease, reflecting the impact of caring responsibilities on labour market outcomes.
For children without access to treatment, it is assumed that caring responsibilities primarily affect a single parent or primary carer, consistent with a conservative modelling approach. Productivity losses for carers are estimated using the same two channels as for patients.
Losses associated with reduced workforce participation are estimated by applying evidence on the likelihood of being out of work among parents of children with a rare disease to the population of carers of children with unmanaged disease. The resulting reduction in employment is valued using average output per worker to estimate lost GVA due to non-participation.
In addition, absenteeism-related productivity losses are estimated for carers who remain in work, using assumptions on the average number of days off work attributable to caring responsibilities, hours worked per day and GVA per hour. These estimates capture the impact of frequent absences and reduced availability for work associated with unmanaged paediatric rare disease.
Total productivity loss for carers is calculated as the sum of workforce participation and absenteeism losses and represents the baseline annual productivity loss associated with caring for a child with unmanaged rare disease.
Summary measure of baseline productivity loss
Across working-age patients and carers, baseline productivity losses associated with unmanaged rare disease are expressed as:
- Total annual GVA lost due to reduced workforce participation and absenteeism; and
- Average productivity loss per unmanaged patient or per carer.
These baseline estimates provide the foundation for subsequent steps in the analysis, which assess the extent to which access to effective treatment reduces productivity losses and scale these gains by the number of additional patients gaining access to treatment under each scenario.
Step 2: Reduction in productivity loss associated with treatment
The second step estimates the extent to which access to effective treatment reduces productivity losses associated with unmanaged disease, applying available estimates from the literature of the reduction in productivity loss associated with moving from unmanaged to managed rare disease.
For patients, evidence from a Chiesi European report is used to estimate the percentage reduction in productivity loss associated with treatment availability. For carers, published US evidence is used, with results adjusted to the UK context by scaling carer productivity improvements in line with observed regional differences in-patient productivity impacts.
For the purposes of this analysis, it is assumed that only one informal carer per patient is affected by caring responsibilities. Any reduction in productivity loss is therefore applied to a single parent or primary carer only. This assumption is intended to be conservative, as it excludes potential productivity impacts on additional carers or wider family members.
Applying these evidence-based reductions to the baseline productivity loss estimates yields an estimate of the reduction in productivity loss per treated patient and per treated carer.
Step 3: Scaling by the number of patients gaining access to treatment
The per-person reductions in productivity loss are then scaled by the incremental number of patients gaining access to treatment under each scenario, relative to the base case, coming from the analysis on patient benefits.
The incremental treated population is derived from:
- The diagnosed rare disease population;
- The proportion of rare diseases for which a treatment exists (treatment discovery) in the base case and in each scenario; and
- The proportion of eligible patients able to access treatment (treatment access) in the base case and in each scenario.
Only working-age patients are included in the productivity analysis. Productivity impacts for carers are scaled consistently using the same incremental treated patient population.
Step 4: Discounting and cumulative productivity benefits
Reductions in productivity loss are monetised using average earnings as a proxy for GVA, producing estimates of avoided productivity losses associated with improved treatment access and discovery.
Productivity benefits are treated as economic (consumption) benefits and are therefore discounted using the HM Treasury Green Book standard discount rate of 3.5 per cent per annum, in contrast to the 1.5 per cent discount rate applied to patient health and life outcomes. This distinction reflects Green Book guidance, which applies the lower discount rate only to health and life outcomes.
As with the patient benefit analysis, the timing of benefit realisation differs by scenario:
- Productivity benefits associated with improved access to existing treatments are assumed to accrue immediately.
- Productivity benefits associated with new treatment discovery are discounted to reflect the time lag between orphan designation, regulatory approval and routine patient access.
In addition to steady-state estimates, the analysis also reports cumulative productivity benefits over ten-year horizons. These horizons are intended to reflect productivity gains that would accrue if patients and carers receive an appropriate intervention at least ten years prior to retirement, capturing benefits over the remaining working life while avoiding assumptions about onset and diagnosis averaged across rare disease, which is outside the scope of this analysis. The analysis takes the productivity benefits per annum as constant and discounts them over a ten-year period. All cumulative productivity benefits are discounted at 3.5 per cent per annum, consistent with the treatment of productivity as an economic outcome.
Table 3 Productivity benefits including rare cancer
| Source | Working-age patients | Carers | Total (£billion) | Source | |
|---|---|---|---|---|---|
| Productivity loss per unmanaged patient | (A) | £48,025 | £36,308 | Survey evidence | |
| % remaining productivity loss | (B) | 51% | 20% | Chiesi, 2022; Chiesi, 2023 † | |
| Productivity loss per managed patient | (C) | £24,406 | £7,260 | (C) = (A)*(B) | |
| Reduction in productivity loss per patient | (D) | £23,619 | £29,047 | (D) = (A) – (C) | |
| Enhanced access scenario | (F) | £0.8 | £0.6 | £1.1 (discounted) | (D)*(Δ treated patients) |
| Best in Europe access scenario | (G) | £1.6 | £1.2 | £2.3 (discounted) | (D)*(Δ treated patients) |
Source: Frontier Economics
Note: † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US).
Annex D Methodology: Valuing impact on Carer's Allowance
This annex sets out the approach used to estimate the public expenditure impacts on Carer's Allowance associated with improved access to rare disease treatments. These impacts reflect reductions in eligibility for Carer's Allowance where treatment reduces caring intensity sufficiently to enable carers to return to work. The analysis focuses on avoided Carer's Allowance payments, valued as direct savings to government expenditure. It is treated separately from productivity impacts to avoid double counting, as Carer's Allowance represents a fiscal transfer rather than an economic productivity effect.
Step 1: Identifying carers eligible for Carer's Allowance
The analysis focuses on parents and informal carers of children living with rare disease, as this group accounts for the majority of Carer's Allowance claims related to rare disease.
The starting point is the number of working-age adults who are out of work due to caring responsibilities for a child with unmanaged rare disease. This population is derived using:
- The number of children diagnosed with rare disease;
- The proportion without access to treatments;
- Evidence on labour market participation among parents of children with rare disease.
Consistent with the productivity analysis, it is assumed that only one parent or primary carer per child is affected by caring responsibilities. This assumption is conservative and excludes impacts on additional carers.
Step 2: Reduction in Carer's Allowance eligibility associated with treatment
Improved access to treatment is assumed to reduce caring intensity for a subset of parents, enabling some carers to return to work and therefore no longer meeting eligibility criteria for Carer's Allowance.
The analysis applies evidence-based estimates of the remaining productivity loss among carers following treatment to infer the proportion of carers who would be able to re-enter employment. This produces an estimate of the number of carers no longer eligible for Carer's Allowance under each scenario relative to the base case.
Step 3: Monetisation and discounting
Avoided Carer's Allowance payments are monetised using the statutory weekly Carer's Allowance rate, applied to the estimated number of carers exiting eligibility under each scenario.
As Carer's Allowance does not represent a health or life outcome, impacts are discounted using the HM Treasury Green Book standard discount rate of 3.5 per cent per annum.
Annex E Results: Additional modelled scenarios
In addition to the core analysis presented in the main report, a range of scenarios were also modelled. These are set out in this Annex alongside the results from the core analysis.
Core scenarios
- Enhanced access. The increase in treatment access from better reimbursement rates on the NHS combined with the expected expansion in licensed treatment availability leads to £2.1 billion of additional QALY benefits compared to the base case, and to additional annual productivity benefits of £1.1 billion (discounted).
- Best in Europe access. Under the pipeline-informed treatment availability assumption, combined with best in Europe access reimbursement rates, additional QALY benefits amount to £4.4 billion, while annual productivity benefits increase to approximately £2.3 billion annually (discounted).
Additional scenarios
- Additional scenario: using mean rather than median lifetime QALY gain. We model an additional scenario where we assume the mean rather than the median lifetime QALY gain (0.99 vs. 0.43 QALYs per treated patient). Our estimates indicate that this would increase lifetime benefits from £4.4 billion to £10.0 billion in the best in Europe access scenario. The mean QALY estimate is higher than the median as it reflects the small number of breakthrough treatments that can generate significant QALY gains. Both the mean and median QALY estimates are taken from the same review, based on the distribution of benefits observed across treatment approvals from 1999 to 2019. The mean may be a reasonable forward-looking perspective if new rare disease treatments are more likely to resemble the higher-impact therapies seen in the historical sample.
- Additional scenario: improvement in reimbursement rates only (no improvement in treatment availability). We model an additional scenario in which reimbursement rates improved while treatment availability remains at current levels (i.e. 5 per cent of rare diseases have a licensed treatment without assuming expansion from the pipeline). Under the assumption that the UK moves to the top three in treatment access, improving treatment access via NHS reimbursement alone leads to additional QALY benefits of £0.4 billion. Assuming, instead, that the UK becomes the best in Europe, additional lifetime QALY benefits amount to £1.3 billion.
- Additional scenario: estimate of benefits to patients and to the economy excluding rare cancers. We have modelled an additional scenario where benefits to patients and to the economy are computed excluding rare cancers from the list of considered conditions. Table 5 and Table 6 report detailed results for estimated benefits to patients and productivity benefits of this scenario.
Table 4 Summary patient and productivity benefit results for all scenarios
| Scenarios | Patient benefits (lifetime Health-Related Quality of Life value), (£billion, discounted) | Productivity benefits (annual), (£billion, discounted) | ||
|---|---|---|---|---|
| Enhanced access | Best in Europe access | Enhanced access | Best in Europe access | |
| Core scenario | £2.1 | £4.4 | £1.1 | £2.3 |
| Adopting mean QALY gain as opposed to median | £4.8 | £10.0 | £1.1 | £2.3 |
| Improvement in reimbursement rates only | £0.4 | £1.3 | £0.2 | £0.8 |
| Core scenario excluding rare cancer | £1.4 | £2.8 | £0.7 | £1.5 |
Source: Frontier Economics
Note: Summary of scenario analysis, estimation steps detailed in more detail in Table 2 and Table 5
Note: Summary of scenario analysis, estimation steps detailed in more detail in Table 2 and Table 5
Table 5 Estimated patient benefits excluding rare cancer (lifetime Health-Related Quality of Life value)
| Item | Base case | Enhanced access | Best in Europe access | Source | |
|---|---|---|---|---|---|
| UK population | (A) | 69,300,000 | ONS | ||
| % diagnosed with rare disease | (B) | 1.50% | Thygesen et al, 2025 | ||
| UK patients diagnosed with rare disease | (C) | 1,039,500 | 1,039,500 | 1,039,500 | C = A*B |
| Treatment exists to manage condition | (D) | 5% | 13% | 13% | UK govt. (base), Fermaglich and Miller, 2023 † (scenario) |
| Patient access (% public availability) | (E) | 35% | 50% | 89% | Patient W.A.I.T Indicators (base and scenario) |
| Patients with diagnosis and treatment | (F) | 18,191 | 67,568 | 120,270 | F = D*E*F |
| QALY benefit with treatment | (G) | 0.43 | 0.43 | 0.43 | Clifford et al, 2024 |
| Total QALYs | (H) | 7,822 | 29,054 | 51,716 | H = F*G |
| Value per QALY: Green Book appraisal value (in £), unadjusted (typical NICE values, Green Book value adjusted) | (I) | £70,000 (£30,000 – £87,500) | |||
| HRQoL value (£billion) | (J) | £0.6 (£0.2 – £0.7) |
£2.0 (£0.9 – £2.6) |
£3.6 (£1.6 – £4.5) |
J = H*I |
| Delta HRQoL value (£billion) | (K) | £1.5 (£0.6 – £1.9) |
£3.1 (£1.3 – £3.9) |
K = Lscenario – Lbase | |
| Delta HRQoL value (£billion, discounted) | (L) | £1.4 (£0.6 – £1.7) |
£2.8 (£1.2 – £3.5) |
Green Book discount rate for health and life outcomes | |
Source: Frontier Economics
Note: Estimates shown according to central QALY value of £70,000, analysis was also run using range of £30,000 – £87,500. † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US)
Note: Estimates shown according to central QALY value of £70,000, analysis was also run using range of £30,000 – £87,500. † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US)
Table 6 Productivity benefits excluding rare cancer
| Source | Working-age patients | Carers | Total (£billion) | Source | |
|---|---|---|---|---|---|
| Productivity loss per unmanaged patient | (A) | £48,025 | £36,308 | Survey evidence | |
| % remaining productivity loss | (B) | 51% | 20% | Chiesi, 2022; Chiesi, 2023 † | |
| Productivity loss per managed patient | (C) | £24,406 | £7,260 | (C) = (A)*(B) | |
| Reduction in productivity loss per patient | (D) | £23,619 | £29,047 | (D) = (A) – (C) | |
| Enhanced access scenario | (F) | £0.5 | £0.4 | £0.7 (discounted) | (D)*(Δ treated patients) |
| Best in Europe access scenario | (G) | £1.0 | £0.8 | £1.5 (discounted) | (D)*(Δ treated patients) |
Source: Frontier Economics
Note: † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US).
Note: † Uses the US jurisdiction definition of rare disease (<200,000 affected in the US).
References
1 DHSC (2021). The UK Rare Diseases Framework.
2 MHRA (2025) Rare therapies and UK regulatory considerations. Accessed December 2025: https://www.gov.uk/government/publications/rare-therapies-and-uk-regulatory-considerations/rare-therapies-and-uk-regulatory-considerations
3 Fermaglich and Miller (2023) A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease; MHRA (2025) Rare therapies and UK regulatory considerations. Accessed December 2025: https://www.gov.uk/government/publications/rare-therapies-and-uk-regulatory-considerations/rare-therapies-and-uk-regulatory-considerations
4 IQVIA, EFPIA (2025). EFPIA Patients W.A.I.T Indicator 2024 survey
5 Lodh R, Hou B, Hough A, Oddie S, Mason D, Wright J. Health care utilisation and education outcomes of children with rare diseases: a born in Bradford cohort study. Eur J Pediatr. 2023;182(12):5511-5517. doi:10.1007/s00431-023-05225-
6 Eurordis (2017) Juggling care and daily life: The balancing act of the rare disease community
7 Fermaglich LJ and Miller KL (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease.
8 DHSC, DSIT (2023). Commercial clinical trials in the UK: the Lord O'Shaughnessy review - final report
9 Mazzucato M, Visonà Dalla Pozza L, Minichiello C, Toto E, Vianello A, Facchin P. (2023). Estimating mortality in rare diseases using a population-based registry, 2002 through 2019. Orphanet Journal of Rare Disease. 18(1): 362. Published 2023 Nov 17. doi:10.1186/s13023-023-02944-7
10 Thygesen JH, Zhang H, Issa H, et al. (2025). Prevalence and demographics of 331 rare diseases and associated COVID-19-related mortality among 58 million individuals: a nationwide retrospective observational study. Lancet Digit Health. 7(2): e145-e156. doi:10.1016/S2589-7500(24)00253-X
11 Dumbuya JS, Zeng C, Deng L, et al. (2025). The impact of rare diseases on the quality of life in paediatric patients: current status. Front Public Health. 13: 1531583. Published 2025 Mar 24. doi:10.3389/fpubh.2025.1531583
12 DHSC (2021). The UK Rare Diseases Framework.
13 DHSC (2025). England Rare Diseases Action Plan 2025: main report.
14 DHSC (2025). England Rare Diseases Action Plan 2025: main report.
15 Fermaglich LJ and Miller KL (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease; MHRA (2025) Rare therapies and UK regulatory considerations. Accessed December 2025: https://www.gov.uk/government/publications/rare-therapies-and-uk-regulatory-considerations/rare-therapies-and-uk-regulatory-considerations
16 IQVIA (2025). EFPIA Patients W.A.I.T Indicator 2024 Survey. Based on full availability of orphan drugs licensed in the EU in England and Scotland.
17 Kamerikar, Dheer, Seoan-Vazquez (2023). HPR78 A Comparison of Orphan Drugs Approved By the FDA and EMA, 1995-2022
18 Lau, F., & Seifert, R. (2026). Comparison of drug approvals of the FDA and EMA between 2013 and 2023. Naunyn-Schmiedeberg's Archives of Pharmacology, 399(1), 279-299.
19 Eurordis, Rare Barometer (2025). The impact of living with a rare disease: Barriers and enablers of independent living and social participation.
20 Eurordis (2017). Juggling care and daily life. The balancing act of the rare disease community.
21 Ibid.
22 DHSC (2021). The UK Rare Diseases Framework.
23 DHSC (2025). England Rare Disease Action Plan 2025: main report.
24 IQVIA, EFPIA (2025). EFPIA Patients W.A.I.T Indicator 2024 survey.
25 Fermaglich LJ and Miller KL (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease.
26 Oancea, C, Gherman, D M, Popescu, FG, Aurelian, SM, Homentcovschi, C (2025). The Uneven Effect of Rare Diseases on Functional Status and Work Capacity. Healthcare. 13(6), 594. https://doi.org/10.3390/healthcare13060594
27 DHSC (2025). England Rare Diseases Action Plan 2025: main report
28 IQVIA, EFPIA (2025). EFPIA Patients W.A.I.T Indicator 2024 survey.
29 IQVIA, EFPIA (2025). EFPIA Patients W.A.I.T Indicator 2024 survey.
30 Yang G et al. The national economic burden of rare disease in the United States in 2019. Orphanet Journal of Rare Diseases (2022)
31 Pennington B et al. Caring for and caring about in economic evaluation: modelling the family and caregiving effects. PharmacoEconomics (2026).
32 DHSC (2025). England Rare Diseases Action Plan 2025: main report.
33 Fit for the Future: 10 Year Health Plan for England.
34 Wakap SN et al. (2019). Estimating cumulative point prevalence of rare diseases: analysis of the Orphanet database.
35 NHSE (2023). Web article. New NHS treatments helping extend survival for babies with rare muscle-wasting disease
36 https://nationalscreening.blog.gov.uk/2025/08/07/uk-nsc-publishes-sma-screening-evidence-documents/
37 Aranda-Reneo et al., 2020. The Burden of Spinal Muscular Atrophy on Informal Caregivers.
38 Parsons et al., 2025. Remaining burden of SMA among treated patients.
39 Eurordis, Rare Barometer (2025). The impact of living with a rare disease: Barriers and enablers of independent living and social participation.
40 Thygesen JH, Zhang H, Issa H, et al. (2025). Prevalence and demographics of 331 rare diseases and associated COVID-19-related mortality among 58 million individuals: a nationwide retrospective observational study. Lancet Digit Health. 7(2): e145-e156. doi:10.1016/S2589-7500(24)00253-X
41 Dumbuya JS, Zeng C, Deng L, et al. (2025). The impact of rare diseases on the quality of life in paediatric patients: current status. Front Public Health. 13:1531583. Published 2025 Mar 24. doi:10.3389/fpubh.2025.1531583
42 Andreu, P., Atay, N. J., Piccinini, E., Chiesi, G., & Cioffi, G. (2023). Rare disease burden of care and the economic impact on citizens in Germany, France and Italy. Published online.
43 Eurordis (2017) Juggling care and daily life: The balancing act of the rare disease community
44 Witt S, Schuett K, Wiegand-Grefe S, Boettcher J, Quitmann J. Living with a rare disease - experiences and needs in pediatric patients and their parents. Orphanet J Rare Dis. 2023;18(1):242. Published 2023 Aug 11. doi:10.1186/s13023-023-02837-9
45 MRA (2025). Press release. Major change for rare disease treatments on the way, signals MHRA
46 Jarius et al., 2023. Update on the diagnosis and treatment of neuromyelitis optica spectrum disorders (NMOSD).
47 Huda et al., 2019. Neuromyelitis optica spectrum disorders.
48 Xue B, Li J, Xie D, et al. Effects of early intervention in neuromyelitis optica spectrum disorder patients with seropositive AQP4 antibodies. Front Immunol. 2024;15:1458556. Published 2024 Nov 1. doi:10.3389/fimmu.2024.1458556
49 Ham AS, Gomez Hjerthen I, Tian D-C, et al. Unemployment, work hour reduction, and income loss: An international, multicentered, cross-sectional study of neuromyelitis optica spectrum disorder. Multiple Sclerosis Journal. 2025;31(11):1358-1369. doi:10.1177/13524585251349139
50 Hughes, D. A., Bourke, S., Jones, A., Bhatt, R., Huda, S., Mutch, K., & Jacob, A. (2022). Health utilities and costs for neuromyelitis optica spectrum disorder. Orphanet Journal of Rare Diseases, 17(1), 159. Published: 07 April 2022. doi: 10.1186/s13023-022-02310-z
51 Frontier Economics (2024). The value of industry clinical trials to the UK – extended report. Accessed February 2026 at: https://www.abpi.org.uk/publications/the-value-of-industry-clinical-trials-to-the-uk-extended-report/
52 Lodh R, Hou B, Hough A, Oddie S, Mason D, Wright J. (2023). Health care utilisation and education outcomes of children with rare diseases: a born in Bradford cohort study. Eur J Pediatr. 182(12): 5511-5517. doi:10.1007/s00431-023-05225-
53 Eurodis (2025). The impact of living with a rare disease: barriers and enablers of independent living and social participation.
54 Lodh R, Hou B, Hough A, Oddie S, Mason D, Wright J. (2023). Health care utilisation and education outcomes of children with rare diseases: a born in Bradford cohort study. Eur J Pediatr. 182(12): 5511-5517. doi:10.1007/s00431-023-05225-
55 Chisei (2023). Rare disease burden of care and the economic impact on citizens in Germany, France, and Italy.
56 Imperial College Health Partners (2018). A preliminary assessment of the potential impact of rare diseases on the NHS.
57 NHS National Cost Collection data 24/25
58 NHS National Cost Collection data 24/25.
59 Navarette-Opazo AA et al. (2021). Can you hear us now? The impact of health-care utilization by rare disease patients in the United States. Genet Med. 23(11): 2194-2201. doi: 10.1038/s41436-021-01241-7.
60 Chung, Claudia CY, et al. "Socio-economic costs of rare diseases and the risk of financial hardship: a cross-sectional study." The Lancet Regional Health – Western Pacific 34 (2023).
61 Nichols, CA., et al. (2025). Characterizing healthcare resource utilization in two rare diseases (Kleefstra syndrome and SLC6A1 epileptic encephalopathy) using multimodal real-world data. Orphanet Journal of Rare Diseases. 20(1): 344.
62 National Institute for Health and Care Excellence. Onasemnogene abeparvovec for treating pre-symptomatic spinal muscular atrophy (MAA partial review of HST 15) [ID4051]: Committee Papers. NICE, 19 Apr. 2023.
63 Zhu C, Zaidman C, Youn B, et al. Evaluation of inpatient and emergency department healthcare resource utilization and costs pre- and post-nusinersen for the treatment of spinal muscular atrophy using United States claims. J Comp Eff Res. Published online July 4, 2024. doi:10.57264/cer-2023-0187
64 Shell, R., Al-Zaidy, S., Arnold, W., Rodino-Klapac, L., Prior, T., Kotha, K., ... & Mendell, J. (2018). SMA THERAPIES I: P. 178AVXS-101 phase 1 gene therapy clinical trial in spinal muscular atrophy type 1: improvement in respiratory and bulbar function reduces frequency and duration of hospitalizations compared to natural history. Neuromuscular Disorders, 28, S82. Published October 2018, doi: 10.1016/j.nmd.2018.06.206
65 Navarette-Opazo AA et al. (2021). Can you hear us now? The impact of health-care utilization by rare disease patients in the United States. Genet Med. 23(11): 2194-2201. doi: 10.1038/s41436-021-01241-7.
66 National Institute for Health and Care Excellence (2025). Garadacimab for Preventing Recurrent Attacks of Hereditary Angioedema in People 12 Years and Over: Final Draft Guidance. Sept. 2025.
67 Knapp et al., 2022. Evaluating the Economic Burden of Relapses in Neuromyelitis Optica Spectrum Disorder: A Real-World Analysis Using German Claims Data
68 Hughes, D. A., Bourke, S., Jones, A., Bhatt, R., Huda, S., Mutch, K., & Jacob, A. (2022). Health utilities and costs for neuromyelitis optica spectrum disorder. Orphanet Journal of Rare Diseases, 17(1), 159. Published: 07 April 2022. doi: 10.1186/s13023-022-02310-z
69 Shell, R., Al-Zaidy, S., Arnold, W., Rodino-Klapac, L., Prior, T., Kotha, K., ... & Mendell, J. (2018). SMA THERAPIES I: P. 178AVXS-101 phase 1 gene therapy clinical trial in spinal muscular atrophy type 1: improvement in respiratory and bulbar function reduces frequency and duration of hospitalizations compared to natural history. Neuromuscular Disorders, 28, S82. Published October 2018, doi: 10.1016/j.nmd.2018.06.206
70 Fermaglich LJ and Miller KL (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease; MHRA (2025). Rare therapies and UK regulatory considerations. Accessed December 2025: https://www.gov.uk/government/publications/rare-therapies-and-uk-regulatory-considerations/rare-therapies-and-uk-regulatory-considerations
71 Fermaglich LJ and Miller KL (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet Journal of Rare Disease.
72 Thygesen JH et al. (2025). Prevalence and demographics of 331 rare diseases and associated COVID-19-related mortality among 58 million individuals: a nationwide retrospective observational study.
73 NHS (2025). Rare Condition Registration Statistics updated to 2022. NHS England. Accessed January 2026: https://digital.nhs.uk/data-and-information/publications/statistical/rare-condition-registration-statistics/updated-to-2022
74 IQVIA (2025). EFPIA Patients W.A.I.T Indicator 2024 Survey. Based on full availability of orphan medicines licensed in the EU in England and Scotland.
75 Clifford KA et al. (2024). The Health benefits, costs, and cost-effectiveness of ultraorphan drugs.
76 Luarini GS et al. (2025). Overview of orphan medicines in European Union: An Analysis of Regulatory and Technical-Scientific Aspects. Note: Uses the US jurisdiction definition of rare disease (<200,000 affected in the US).
77 IQVIA (2025). EFPIA Patients W.A.I.T Indicator 2024 Survey. Based on full availability of orphan medicines licensed in the EU in England and Scotland.
-
KeywordsRare diseases
-
PublisherABPI, Frontier Economics
-
Last modified01 April 2026
-
Last reviewed01 April 2026




